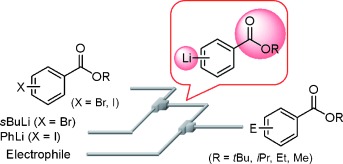
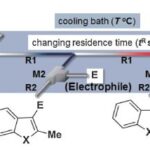
A Flow Microreactor System Enables Organolithium Reactions without Protecting Alkoxycarbonyl Groups.
Nagaki, A.; Kim, H.; Moriwaki, Y.; Matsuo, C.; Yoshida, J.
Chem. Eur. J. 2010, 16, 11167–11177.
DOI: 10.1002/chem.201000876
Generation and reactions of aryl lithiums without protection of ester moieties were achieved.